
Navigating the complexities of COVID-19
As the COVID-19 pandemic continues to impact patients, healthcare systems, and global economic outlooks, we focus on the key considerations and strategies for pharma companies when the data generation for possible therapies is becoming increasingly complex to analyze.
Please note: COVID-19’s impact on pharma is continually evolving. All insights expressed here were formulated based on the situation as of May 26, 2020.
COVID-19 – tracking the fast-moving landscape
There are several COVID-19 treatment and vaccine databases containing an aggregation of publicly-available information from validated sources detailing treatments and vaccines currently in development.
COVID-19 treatments and vaccines (combined)
0
treatments in consideration
0
vaccines in development
Source: Milken Institute
When assessing possible COVID-19 treatment candidates we need to identify the relevant data to collect…
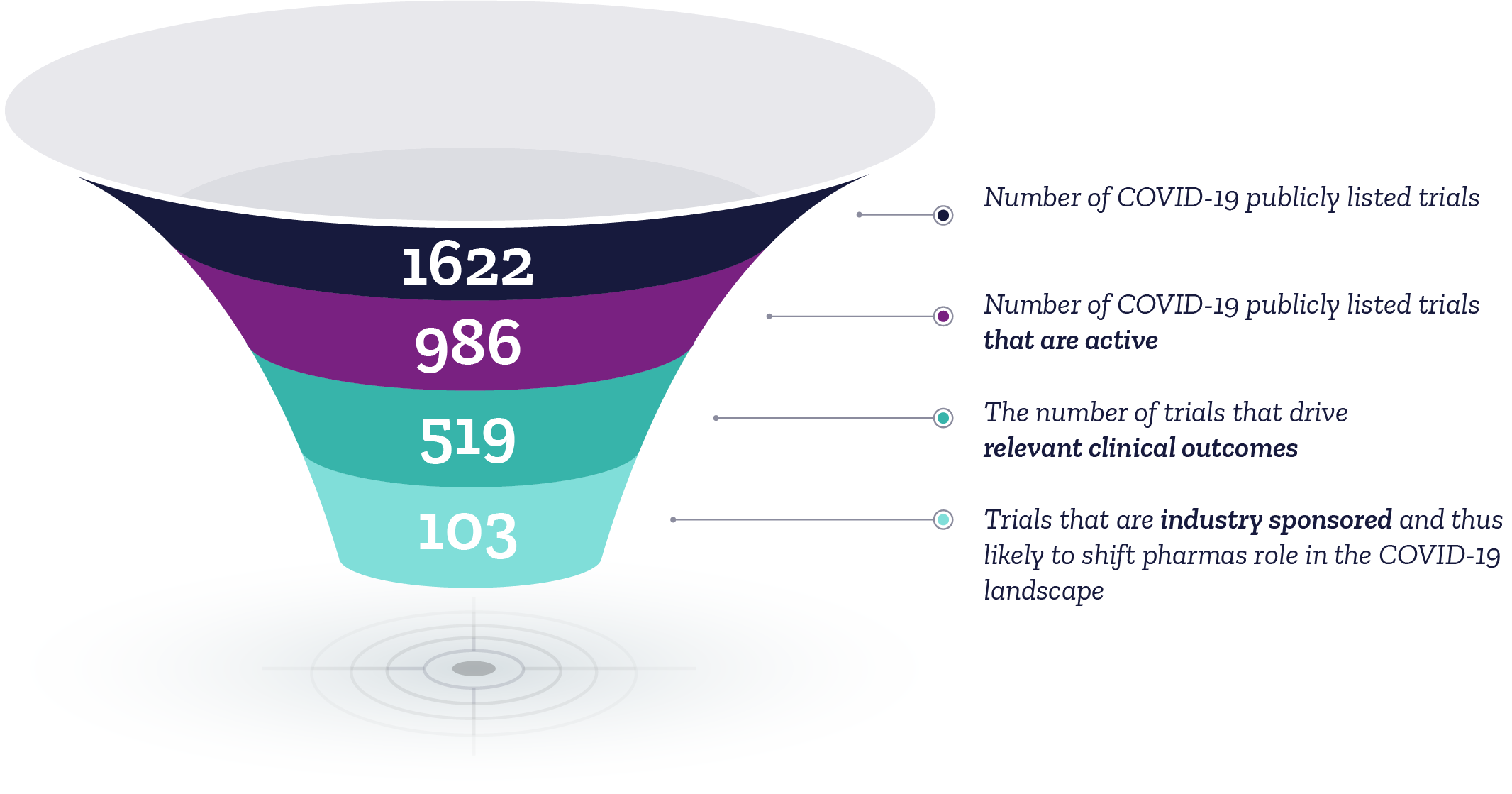
Source: ClinicalTrials.gov
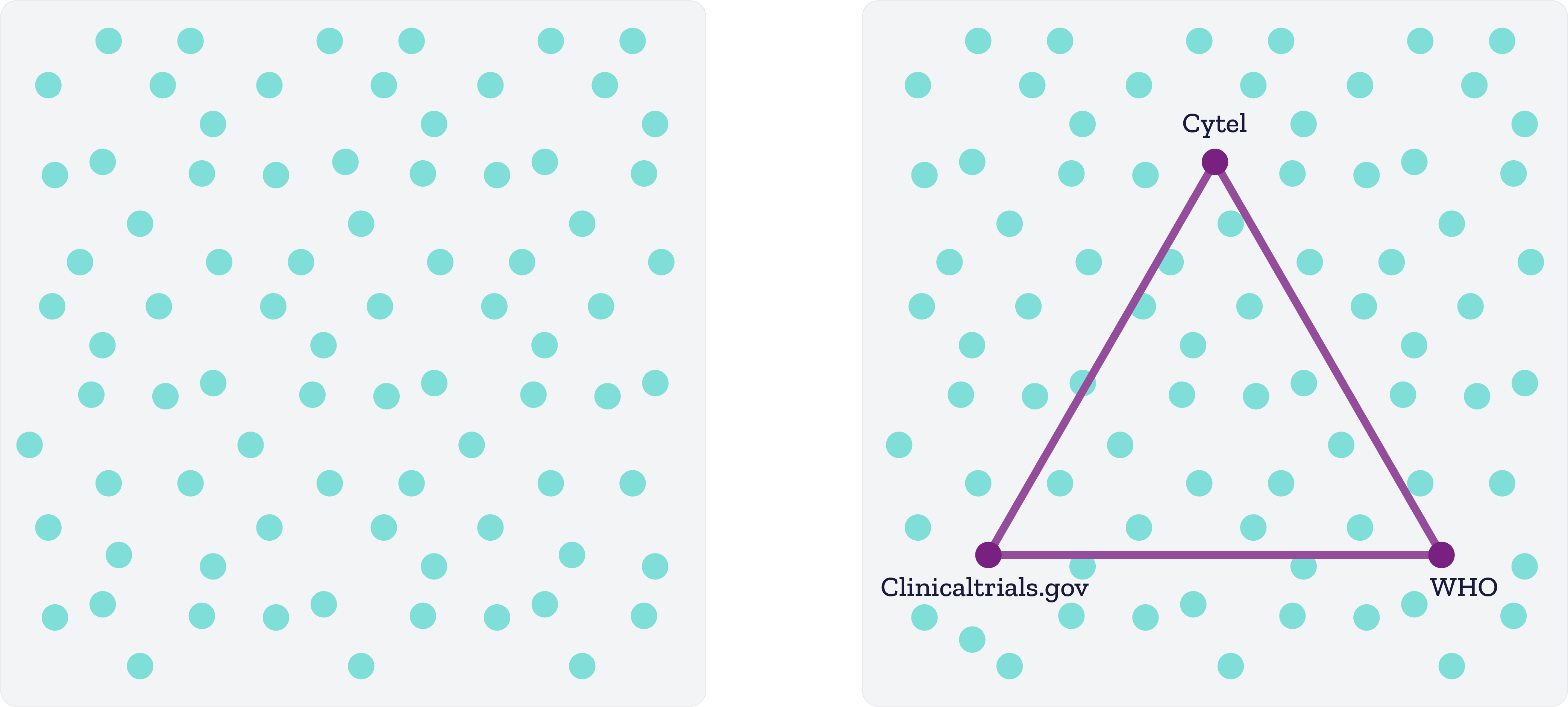
But there is no one true source of global COVID statistics
Two crucial dimensions of data quality are timeliness and accuracy – however there is no one true source of global COVID-19 statistics. We have access to multiple information streams which are often contradictory in nature.
Once we have confidence our data is relevant and accurate we need to ensure we draw meaningful conclusions
Top 12 COVID-19 treatments ranked by number of clinical trials compared to the number of patients
Licensed v experimental COVID-19 treatments – comparing the number of trials and patients
Number of patients
Number of trials
¹ Source: Biomedtracker
